An Introduction to Heat Pumps
A heat pump is a device that is primarily used to transfer heat from a lower ambient temperature into a higher temperature system. Its function is, in essence, pumping heat from one place to another, that is why it is called a ‘heat pump’. This is in contrast to air-conditioners and refrigerators whose main function is to extract heat from a cooler system and release it into a warmer surrounding.
It can also be configured such that it can work in either direction to provide both heating and cooling, depending on the need. These are called reversible heat pumps. If the heat pump is dedicated to heating applications only, the term ‘dedicated heat pump’ is sometimes used to differentiate it from reversible air-conditioning units.
In Australia where the climate is considerably varied and where there are four seasons across most of the country, heat pumps are generally used in space heating/cooling and hot water system applications. In residential and commercial establishments, they can be used for air-conditioning, cooling potable water, space heating, and heat pump hot water systems for bathing/sanitation. In Australia, heat pump water heaters (HPWH) comprise approximately 3% of water heaters in use.
Aside from the above-mentioned, heat pumps can also be used in industrial applications such as process cooling/heating, boiler feed water preheating, drying, pasteurization, and washing processes. The advantage of using heat pumps in these applications is that industrial plants have waste heat flows. Instead of releasing this heat into the environment, it is usually more economical to utilize it in some of their step processes.
The diagram shows an example of a water to water heat pump system that recovers heat from various sources to provide increased COP and reduced energy cost.
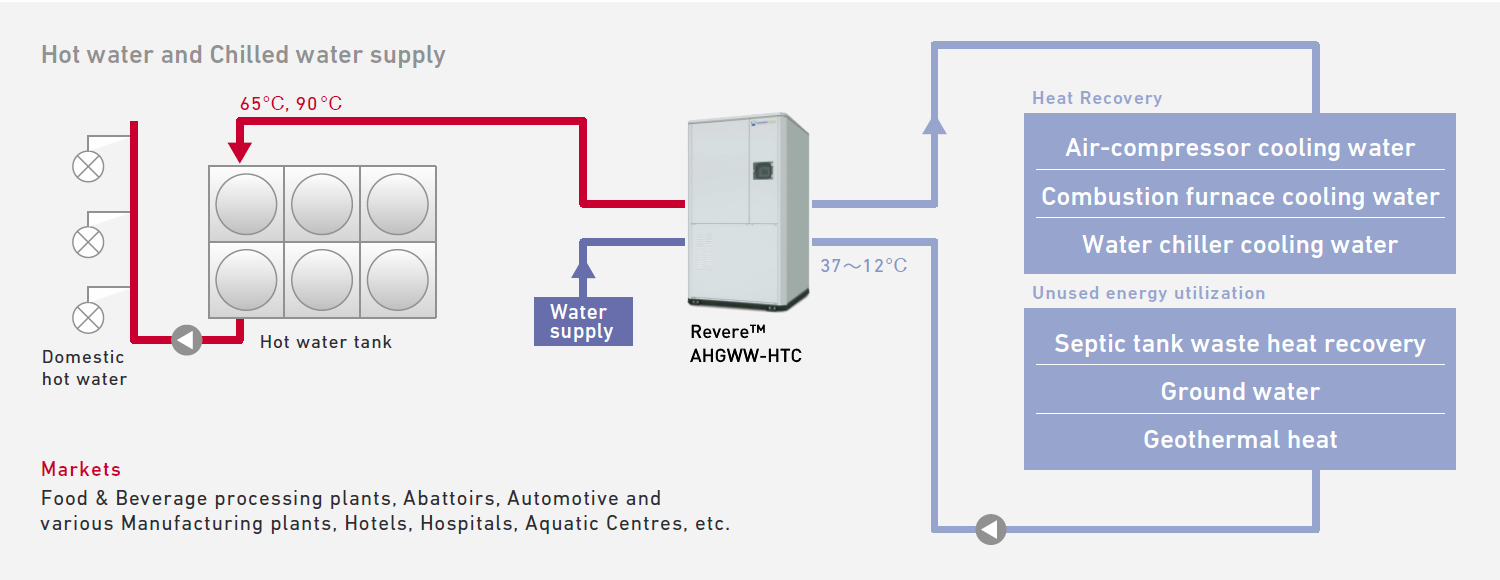

Revere® CO2 Water to Water Heat Pump is a heat pump system that generates hot water and chilled water or hot water only by recovering energy from renewable and waste heat sources such as waste water from industrial processes, cooling water from a conventional cooling tower or ground water/geothermal heat.




